Just Published in Inorganic Chemistry
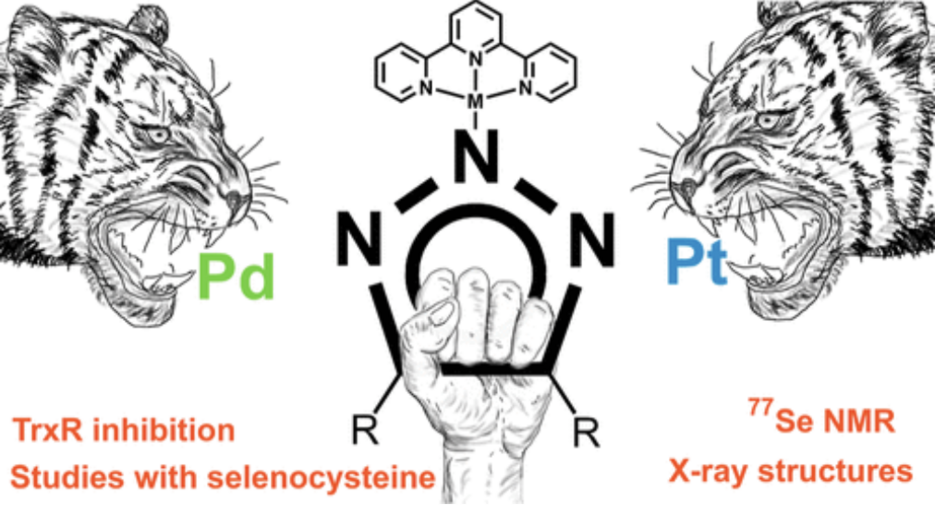
10.07.2023Taming the Biological Activity of Pd(II) and Pt(II) Complexes with Triazolato “Protective” Groups: 1H, 77Se Nuclear Magnetic Resonance and X-ray Crystallographic Model Studies with Selenocysteine to Elucidate Differential Thioredoxin Reductase Inhibition
Authors: Victoria V. L. Müller, Peter V. Simpson, Kun Peng, Uttara Basu, Dominik Moreth, Christoph Nagel, Sebastian Türck, Luciano Oehninger, Ingo Ott, and Ulrich Schatzschneider*
Abstract: The biological activity of Pd(II) and Pt(II) complexes toward three different cancer cell lines as well as inhibition of selenoenzyme thioredoxin reductase (TrxR) was modulated in an unexpected way by the introduction of triazolate as a “protective group” to the inner metal coordination sphere using the iClick reaction of [M(N3)(terpy)]PF6 [M = Pd(II) or Pt(II) and terpy = 2,2′:6′,2″-terpyridine] with an electron-poor alkyne. In a cell proliferation assay using A549, HT-29, and MDA-MB-231 human cancer cell lines, the palladium compound was significantly more potent than the isostructural platinum analogue and exhibited submicromolar activity on the most responsive cell line. This difference was also reflected in the inhibitory efficiency toward TrxR with IC50 values of 0.1 versus 5.4 μM for the Pd(II) and Pt(II) complexes, respectively. UV/Vis kinetic studies revealed that the Pt compound binds to selenocysteine faster than to cysteine [k = (22.9 ± 0.2)·10–3 vs (7.1 ± 0.2)·10–3 s–1]. Selective triazolato ligand exchange of the title compounds with cysteine (Hcys) and selenocysteine (Hsec)─but not histidine (His) and 9-ethylguanine (9EtG)─was confirmed by 1H, 77Se, and 195Pt NMR spectroscopy. Crystal structures of three of the four ligand exchange products were obtained, including [Pt(sec)(terpy)]PF6 as the first metal complex of selenocysteine to be structurally characterized.
Link: https://pubs.acs.org/doi/10.1021/acs.inorgchem.3c02701