Catalytic Formation of Element—Element Bonds
Research
Catalytic Formation of Element—Element Bonds
Transition metal-catalyzed and organocatalytic reactions are indispensable components in the toolbox of organic chemistry. Catalytic methods for the formation of bonds between inorganic elements are significantly less well developed.
Electrophilic activation of difunctional aminoboranes: B–N coupling versus intramolecular Cl/Me exchange
M. Maier, J. Klopf, C. Glasmacher, F. Fantuzzi, J. Bachmann, O. Ayhan, A. Koner, B. Engels,* H. Helten,* Electrophilic activation of difunctional aminoboranes: B–N coupling versus intramolecular Cl/Me exchange, Chem. Commun. 2022, 58, 4464–4467 [DOI]
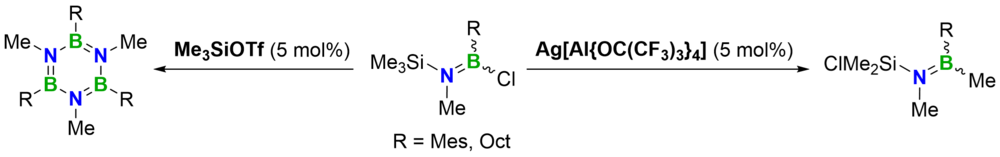
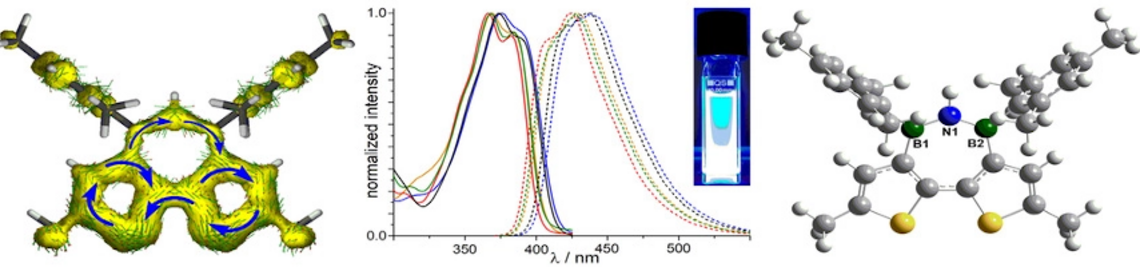
Treatment of an N-silyl-B-chloro-aminoborane with substoichiometric quantities of Me3SiOTf afforded B–N coupling, whereas activation with 5 mol% of Ag[Al{OC(CF3)3}4] led to Cl/Me exchange between the boron and the silicon center. Combined experimental and computational studies of the latter process support a chain reaction that is initiated by nucleation-limited chloride abstraction.
Catalytic B—C Coupling by Si/B Exchange: A Versatile Route to π-Conjugated Organo-borane Molecules, Oligomers, and Polymers
A. Lik, L. Fritze, L. Müller, H. Helten, J. Am. Chem. Soc. 2017, 139, 5692—5695 [DOI].
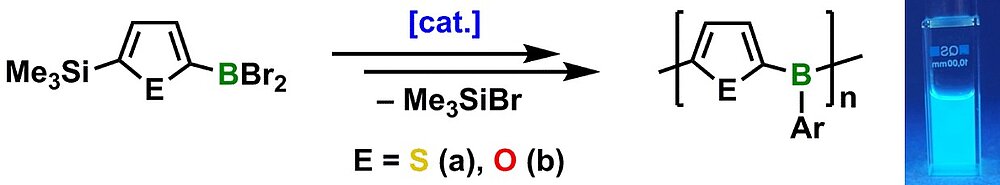
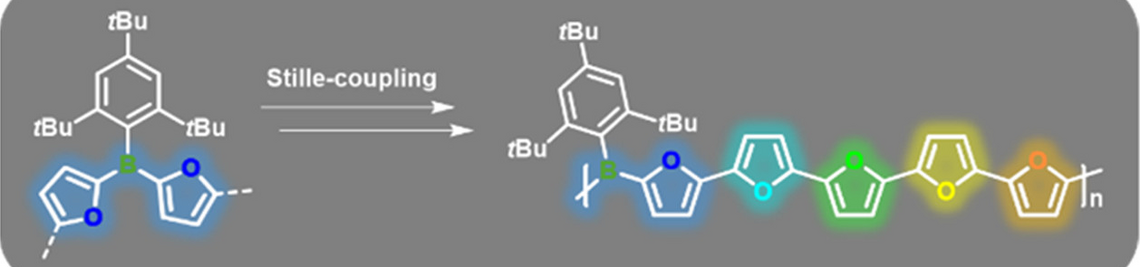
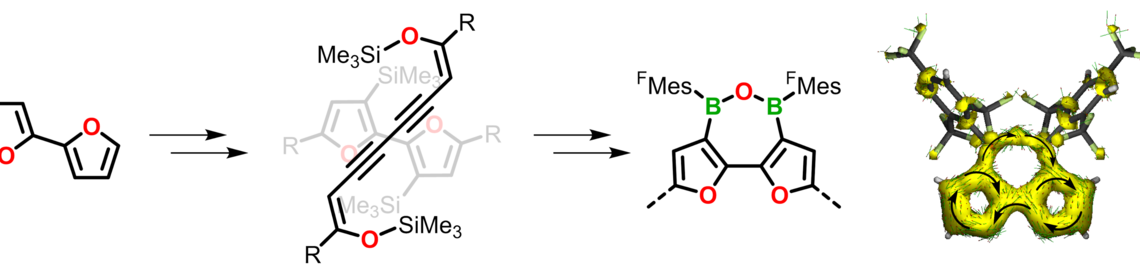
We have developed a highly efficient and environmentally benign B—C bond formation method based on an organocatalytic Si/B exchange reaction, which proved to be a general route to donor—acceptor type conjugated organoborane polymers. Compared to previous routes, our approach uses less toxic and more cost-effective starting materials. Moreover, in some cases the polycondensation may be performed as an AB type process, in which a difunctional monomer is isolated and subsequently polymerized in a controlled manner. Such approaches are extremely desirable as their success is not dependent on the exact stoichiometry of two components.
A. Lik, S. Jenthra, L. Fritze, L. Müller, K.-N. Truong, H. Helten, Chem. Eur. J. 2018, 24, 11961—11972 [DOI] ("Very Important Paper").
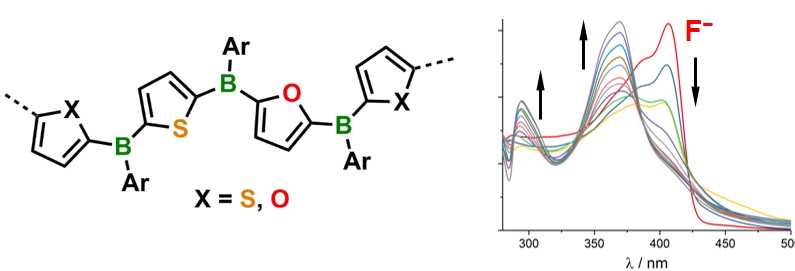
Using this new method, we have prepared a series of arylborane molecules, oligomers, and polymers. Through variation of the furan-to-thiophene ratio, the photophysical properties of these materials are effectively modulated. They are furthermore attractive targets for chemical sensory applications. Binding of fluoride anions is effectively signaled by an optical response.