Molecular Hybrid Materials
Research
Molecular Hybrid Materials
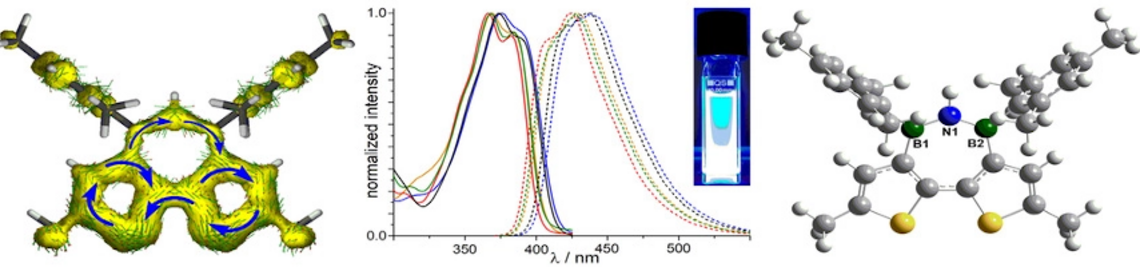
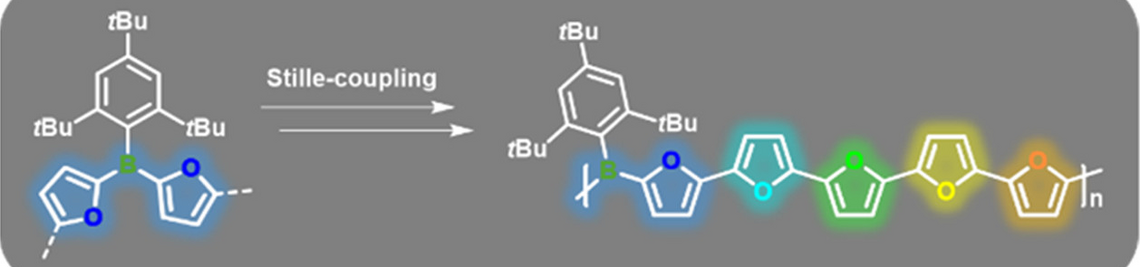
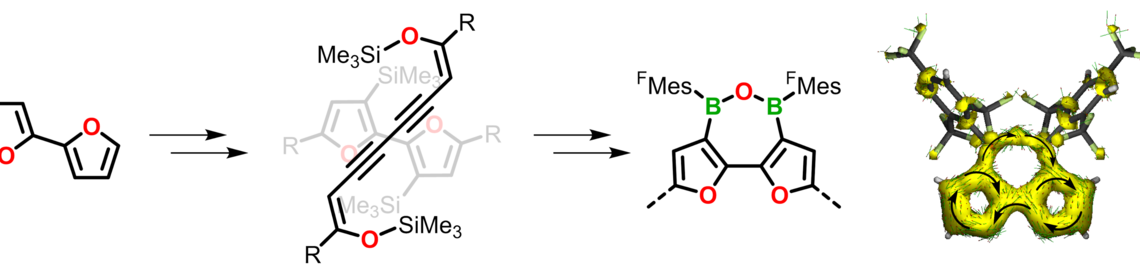
Molecular materials have proven to be versatile alternatives to polymers for the use as photo- or electro-active components in various applications ranging from organic photovoltaics (OPV) to photocatalytic hydrogen production. We are interested in the development of novel molecular organic—inorganic hybrid compounds with specifically tailored electronic and optical properties. Of particular interest is the study of energy and charge transfer processes.
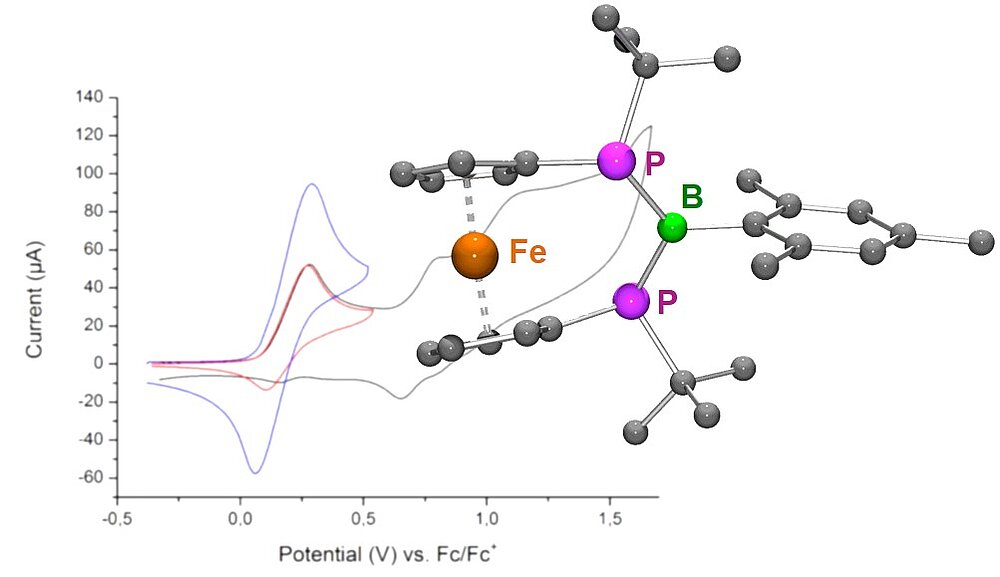
PBP Bridged [3]Ferrocenophane: A Bisphosphanylborane With a Redox Trigger
We prepared an unprecedented P—B—P bridged [3]ferrocenophane in which a bisphosphanylborane fragment (formally valence isoelectronic to the allylic anion) with moderate π interaction is combined with a redox-active ferrocene unit. Two synthetic routes were applied which both provided stereospecific access to the rac diastereomer, contrasting the situation found for other P—E—P bridged [3]ferrocenophanes. Upon electrochemical oxidation, the mono-cation formed undergoes a fast intramolecular electron transfer process from the P—B—P bridge to the ferrocenium moiety, triggered by pyramidal inversion of the P centers. This behaviour is in marked contrast to that of acyclic ferrocenylphosphanes. The lifetime of the initially formed mono-cation has been determined using a rotating ring-disk electrode.