DFT Calculations
Computational chemistry
Modern theoretical electronic structure methods, in particular those based on density functional theory (DFT), allow facile optimization of geometries and calculation of a wide range of molecular properties. Although not involved in method development, the Schatzschneider group routinely applies theoretical calculations to assist in the interpretation of experimental data and guide the synthesis of novel molecules by prediction of molecular properties.
Due to its facile use, high performance, and ability to predict a large number of spectroscopic parameters at different levels of theory, most of our work uses the ORCA program package from the group of Prof. Dr. Frank Neese at the Max-Planck-Institute for Chemical Energy Conversion (MPI CEC) in Mülheim/Ruhr.
Calculations are run on the Linux Cluster of the Leibniz Supercomputing Centre (LRZ) in Munich. Below, there is an overview of current and past applications of DFT calculation in our research.
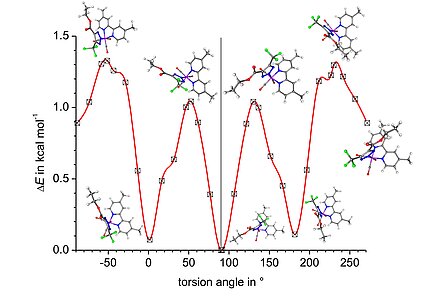
Conformational preferences
In addition to the metal-ligand interactions in the inner coordination sphere of our metal complexes, we are particularly also interested in the orientation of functional groups at the outer ligand periphery, which are important to rationalise the interaction of such systems with bio(macro)molecules. DFT calculations are a facile means to study the conformational preference of organometal and coordination compounds and ORCA offers an easy way to map the energy hypersurface of given molecules.
- P. Schmid, M. Maier, H. Pfeiffer, L. Henry, A. Belz, A. Friedrich, F. Schönfeld, K. Edkins, U. Schatzschneider, Catalyst-free room-temperature iClick reaction of molybdenum(II) and tungsten(II) azide complexes with electron-poor alkynes: Structural preferences and kinetic studies, Dalton Trans. 46, 13386-13396 (2017)
- L. Henry, C. Schneider, B. Mützel, P.V. Simpson, C. Nagel, K. Fucke, U. Schatzschneider, Amino acid bioconjugation via iClick reaction of an oxanorbornadiene-masked alkyne with a MnI(bpy)(CO)3-coordinated azide, Chem. Commun. 50, 15692-15695 (2014)
- S.I. Kirin, U. Schatzschneider, D. Köster, D. Siebler, N. Metzler-Nolte, A systematic evaluation of different hydrogen bonding patterns in unsymmetrical 1,n’-disubstituted ferrocenoyl peptides, Inorg. Chim. Acta 362, 894-906 (2009)
- S.I. Kirin, U. Schatzschneider, X. de Hatten, T. Weyhermüller, N. Metzler-Nolte, 1,n'-Disubstituted ferrocenoyl peptides: Conformational analysis by CD spectroscopy, X-ray crystallography, and DFT calculations, J. Organomet. Chem. 691, 3451-3457 (2006)
UV/Vis and vibrational spectroscopy
Vibrational band positions and UV/Vis electronic excitations can be calculated with high accuracy using DFT and TDDFT methods. In our current work, we particularly utilize such calculations to rationalise the factors which govern efficient photoinduced ligand release in our small-signalling molecules project and the assignment of intermediates in the CO release from metal carbonyl complexes by comparison with the vibrational signature of transient species observed in time-resolved pump-probe spectroscopy.
- C. Nagel, S. McLean, R.K. Poole, H. Braunschweig, T. Kramer, U. Schatzschneider, Introducing [Mn(CO)3(tpa-k3N)]+ as a novel photoactivatable CO-releasing molecule with well-defined iCORM intermediates - Synthesis, spectroscopy, and antibacterial activity, Dalton Trans. 43, 9986-9997 (2014)
- P. Govender, S. Pai, U. Schatzschneider, G. Smith, Next generation PhotoCORMs: Polynuclear tricarbonylmanganese(I)-functionalised polypyridyl metallodendrimers, Inorg. Chem. 52, 5470-5478 (2013)
- P. Rudolf, F. Kanal, J. Knorr, C. Nagel, J. Niesel, T. Brixner, U. Schatzschneider, P. Nürnberger, Ultrafast photochemistry of a manganese-tricarbonyl CO-releasing molecule (CORM) in aqueous solution, J. Phys. Chem. Lett. 4, 596-602 (2013)
Mössbauer parameters
The 57Fe Mössbauer isomer shift and quadrupole splitting is a very sensitive indicator of the number of d electrons, spin state, and coordination environment of the metal center. In a recent collaboration with the group of Ulf-Peter Apfel at the Ruhr-Universität Bochum, we have calculated these parameters using DFT for a range of dinitrosyl iron complexes (DNICs). An excellent agreement of calculated values with experimental data was found for the isomer shift, while the quadrupole splitting was more difficult to model accurately. Such compounds also have relevance in the context of small molecule signalling.
- F. Wittkamp, C. Nagel, P. Lauterjung, B. Mallick, U. Schatzschneider, U.-P. Apfel, Phosphine-ligated dinitrosyl iron complexes for redox-controlled NO release, Dalton Trans. 45, 10271-10279 (2016)
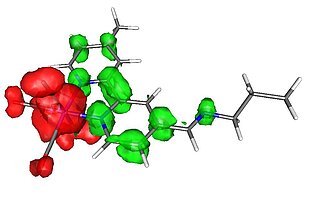
Spin densities and exchange coupling constants
The interest of Prof. Schatzschneider in stable radical ligands can be traced back to his doctoral thesis project in the group of Prof. Dr. Eva Rentschler, at that time at the Max-Planck-Institute for Bioinorganic Chemistry in Mülheim/Ruhr. Since then, we have continued to utilized DFT calculations for an insight in the spin density distribution in open-shell molecules and the calculation of the exchange coupling parameter J indicative of either ferro- or antiferromagnetic interactions in multi-spin systems.
- A.-M. Pütz, U. Schatzschneider, E. Rentschler, Integrated experimental and computational spectroscopy study on the protonation of the alpha-nitronyl nitroxide radical unit, Phys. Chem. Chem. Phys. 14, 1649-1653 (2012)
- M. Jung, A. Sharma, D. Hinderberger, S. Braun, U. Schatzschneider, E. Rentschler, Long-distance magnetic interaction of exchange-coupled copper dimers with nitronyl nitroxide and tert-butyl-nitroxide radicals, Inorg. Chem. 48, 7244-7250 (2009)
- M. Jung, A. Sharma, D. Hinderberger, S. Braun, U. Schatzschneider, E. Rentschler, Nitronyl nitroxide radicals linked to exchange-coupled metal dimers - Studies using X-ray crystallography, magnetic susceptibility measurements, EPR spectroscopy, and DFT calculations, Eur. J. Inorg. Chem. 1495-1502 (2009)
- N. Roques, P. Gerbier, U. Schatzschneider, J.-P. Sutter, P. Guionneau, D. Luneau, E. Rentschler, C. Guérin, Experimental and theoretical studies on magnetic exchange in silole-bridged diradicals, Chem. Eur. J. 12, 5547-5562 (2006)
- C. Rancurel, H. Heise, F. Köhler, U. Schatzschneider, E. Rentschler, J. Vidal, J. Veciana, J.-P. Sutter, Spin transfer and magnetic interaction via phosphorous in nitronyl nitroxide radical-substituted triphenylphosphine derivatives, J. Phys. Chem. A 108, 5903-5914 (2004)
- U. Schatzschneider, E. Rentschler, Electronic and conformational influences on the spin density distribution in substituted tert-butyl phenyl nitroxides, J. Mol. Struct. THEOCHEM 638, 163-168 (2003)
- U. Schatzschneider, T. Weyhermüller, E. Rentschler, Metal complexes with nitronyl nitroxide substituted phenolate ligands providing new magnetic exchange interaction pathways - Synthesis, structures, magnetic dilution studies and ab initio calculations, Eur. J. Inorg. Chem. 2569-2586 (2001)