Inorganic Main Group Macromolecules
Research
Inorganic Main Group Macromolecules
Polymers with a backbone comprised of inorganic elements often show useful properties and functions that complement those of state-of-the-art organic macromolecular materials. Prominent examples are the well-known silicones (polysiloxanes), which are nowadays regarded as commodity products.
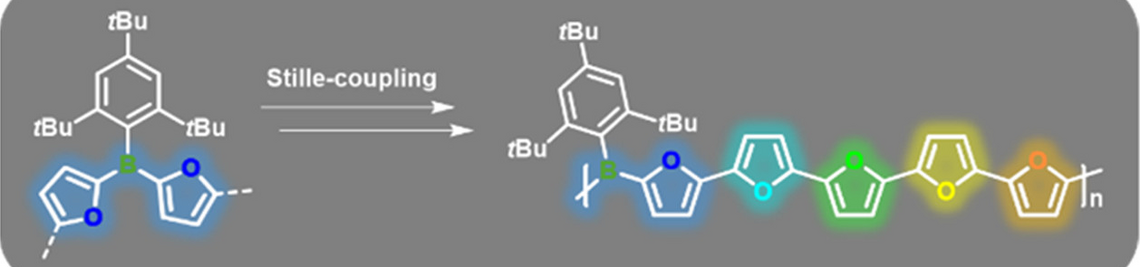
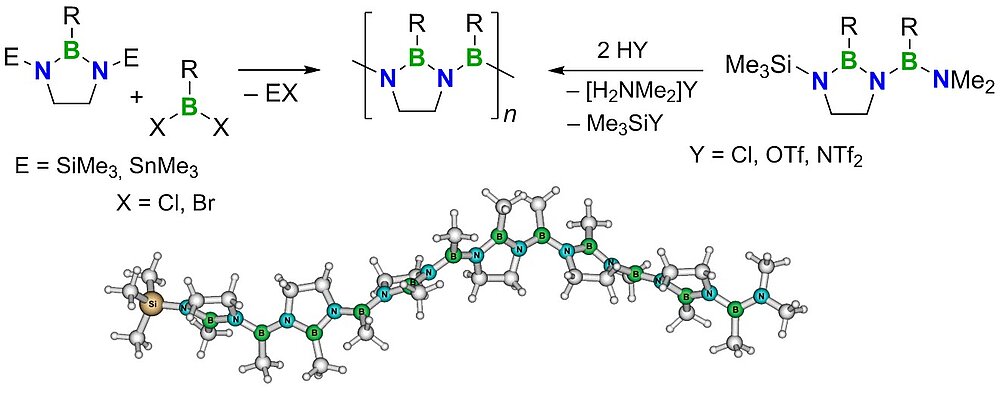
Boron—nitrogen polymers, on the other hand, are significantly less well explored. Poly(aminoborane)s (PABs) became accessible only quite recently through the work by Manners and co-workers. Surprisingly, the unsaturated poly(iminoborane)s (PIBs), which are formally isoelectronic to polyacetylene, have not been convincingly characterized thus far. The failure of previous attempts to prepare such species from rational monomers was ascribed to competing side reactions yielding cyclic iminoborane oligomers, in particular, the cyclic trimer, i.e. borazine, which is a relatively stable benzene analogue.
Linear and macrocyclic oligo(p-phenylene iminoboranes) with ferrocenyl side groups – observation of selective, non-templated macrocyclization
J. S. Schneider, I. Krummenacher, H. Braunschweig, H. Helten,* Linear and macrocyclic oligo(p-phenylene iminoboranes) with ferrocenyl side groups – observation of selective, non-templated macrocyclization, Chem. Commun. 2023 [DOI]
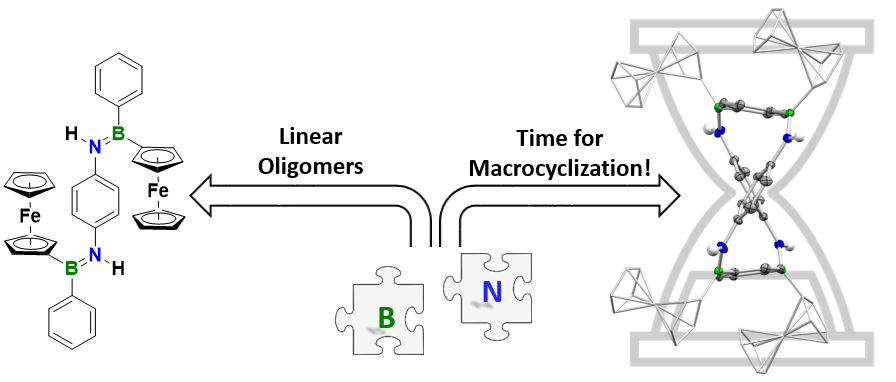
Linear and macrocyclic oligo(phenylene iminoboranes) with ferrocenyl side groups have been prepared, selective macrocyclization proceeds without a template.
Poly(iminoborane)s: The Missing Link in Inorganic Main Group Macromolecular Chemistry
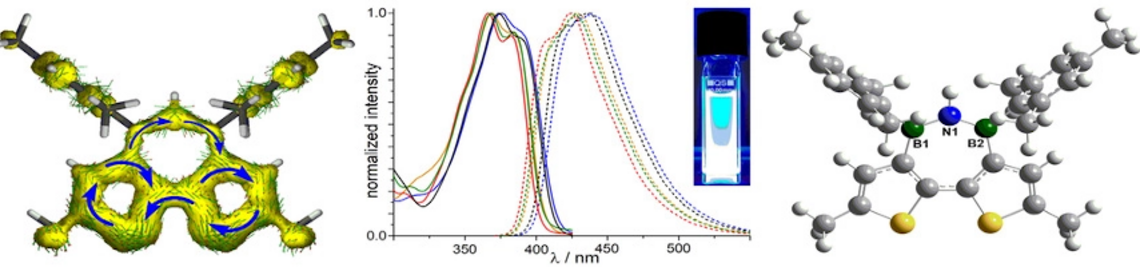
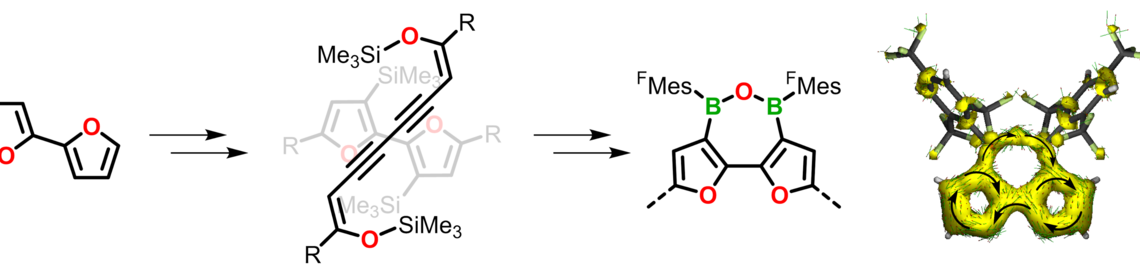
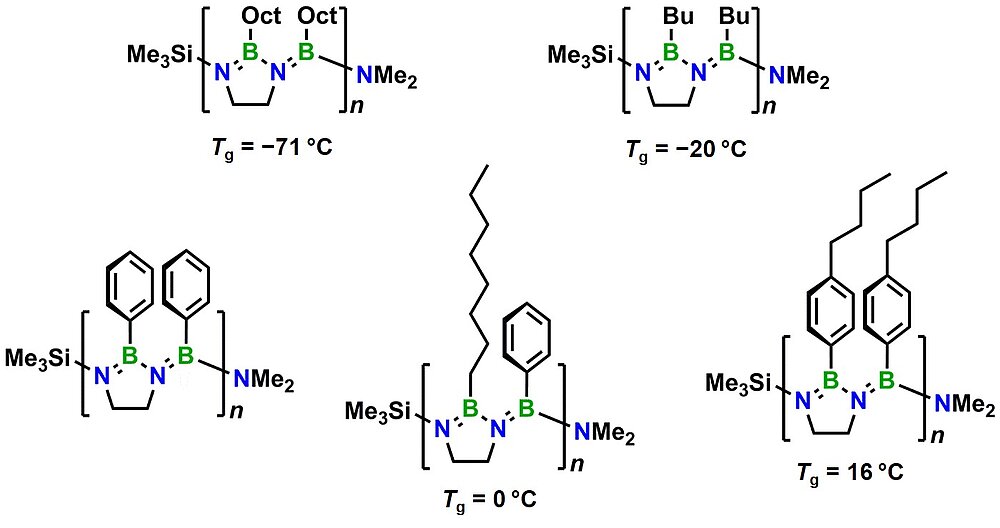
Using an approach that effectively prevents the formation of unwanted cyclic products, we succeeded in the synthesis of the first oligo- and poly(iminoborane)s with a well-defined microstructure. With up to 18 catenated BN units on average, these species comprise the longest BN chains to date. They adopt helical conformations in solution.
The properties of the new materials are effectively tuned by variation of their side groups.