Diamond to Fight Climate Change
09/21/2016Slashing carbon dioxide emissions in an eco-friendly way and producing valuable raw materials in the process: This is the goal of a new Germany-wide research venture. Professor Anke Krüger from Würzburg is on board the project.
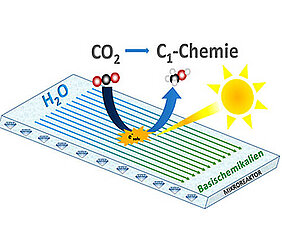
Take a diamond with complex organic molecules on its surface, light of a specific wavelength and add water and carbon dioxide: These are the ingredients of a new microreactor set to improve humanity's carbon footprint.
A new, national research association involving Fraunhofer Institute for Chemical Technology - Institute for Microtechnology Mainz (ICT-IMM), Sahlmann Photochemical Solutions and the University of Würzburg are working together to develop this ground-breaking system. The project titled CarbonCat will be funded with around EUR 1.36 million by the Federal Ministry of Education and Research over the next three years. Anke Krüger, a Würzburg professor of organic chemistry, is in charge of engineering the diamond surfaces.
Need to reduce carbon emissions
Background info: Carbon dioxide (CO2) is a greenhouse gas that is considered to be the major cause of global warming. According to preliminary figures, global CO2 emissions last year totalled 32.1 billion tonnes which is way too high in order to achieve the goals set by the UN Climate Conference in Paris last December. The conference had voted to limit global warming to well below two degrees Celsius compared to pre-industrial levels. To achieve this, greenhouse gas emissions worldwide have to be reduced to zero between 2045 and 2060; and subsequently part of the previously emitted carbon dioxide will have to be removed again from the atmosphere. CarbonCat aims to help accomplish this ambitious target.
Photosynthesis in a technical system
For this purpose, the scientists want to imitate the principle of photosynthesis in a technical system. Instead of plant cells and their photosynthetically active chloroplasts they use a newly developed microreactor which contains a diamond photocatalyst as its photoactive centre. "The special make-up of the microreactor enables the continuous mixing of CO2 and water when irradiated with visible light," explains Thomas Rehm, Senior Scientist at Fraunhofer ICT-IMM and coordinator of the joint project. CarbonCat aims to prove that the approach will enable carbon dioxide to be converted into valuable chemicals such as methanol under near-natural conditions.
Key role for diamond
This makes the chemical optimisation of diamond as a photocatalyst a priority for Anke Krüger besides the technological aspects. "The selective functionalisation of diamond surfaces using complex organic molecules is not trivial, especially regarding the long-term stability for use in a continuous process as we intend to do in the microreactor," she says.
This is a research area the chemist has sufficient experience in: Since last year, she has been coordinating the international research association DIACAT - Diamond materials for the photocatalytic conversion of CO2 to fine chemicals and fuels using visible light. This project also focuses on converting CO2 with the help of diamond, but it takes a different functional approach. "While DIACAT attempts to harvest sunlight particularly through diamond nanostructuring, our focus at CarbonCat is to study both the continuous implementation in a flow reactor and the selective binding of different photoactivation molecules," Krüger explains.
Selective use of light
In addition to the reactor technology and the catalytically active surfaces, both the selection and mix of the required wavelengths and the alignment of the LEDs are essential. "We pay great attention to the interaction of the light source and other system components. This is as important for the photocatalytic process as it is for the overall reactor efficiency," says Benjamin Sahlmann, who is involved in the project as a freelance chemist with his company Sahlmann Photochemical Solutions.
"We hope that the findings gleaned from CarbonCat will allow us to make a contribution to mitigating the environmental impacts resulting from the present CO2 emissions," Thomas Rehm concludes.
The contribution of the project partners
Based on its expertise in developing and testing micro-structured reactors, Fraunhofer ICT-IMM will implement a continuously operated reactor facility that will have the novel diamond photocatalyst at its core. Other tasks of ICT-IMM include the physical adaptation of the diamond material used in the microreactor and a detailed examination of the photocatalytic process during continuous operation.
The working group of Professor Anke Krüger at the University of Würzburg has been studying the production, characterisation and application of nano-scale carbon materials, especially diamond, for more than ten years. The methods for highly stable linking of functional molecules to diamond surfaces developed by the working group are utilised in CarbonCat to optimise the diamond material for being used as a photocatalyst in the microreactor.
Sahlmann Photochemical Solutions will design the light sources for the photocatalysis in the reaction systems for CarbonCat. This task includes manufacturing the customised light sources, their spectral measurement and assessing them in terms of occupational safety.
Contact
Prof. Dr. Anke Krueger, Institute of Organic Chemistry, University of Würzburg
phone +49 931 31-85334, anke.krueger@uni-wuerzburg.de