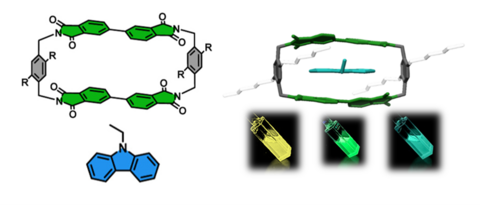
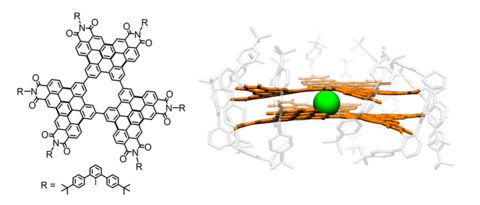
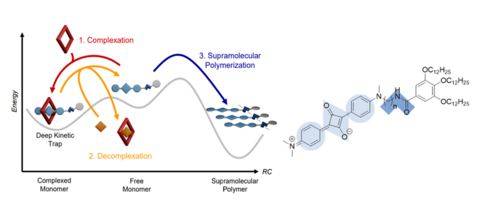
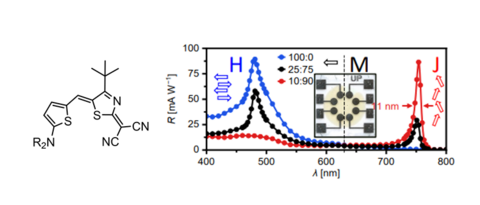
The research programme of our group
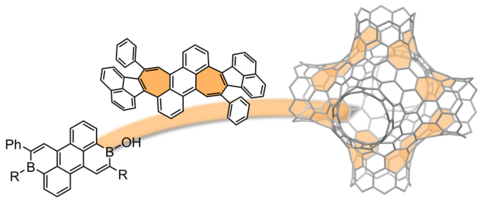
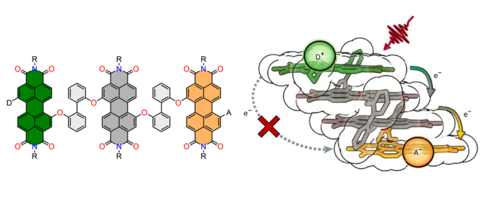
Our research is devoted to the development of advanced organic materials based on functional dyes such as perylene as well as other aromatic imides, merocyanines, squaraines, and boron-doped as well as curved polycyclic aromatic hydrocarbons by employing supramolecular approaches. Towards this objective, we design and synthesize novel molecular building blocks and explore their self-assembly into nanoscale architectures in solution as well as in the liquid-crystalline or solid phase for application in (opto-)electronic and photovoltaic devices as well as in photocatalytic water splitting.