Current Topics
Current Research Topics
Star-shaped Hybrid Materials - Donor-Acceptor Dyades and Triades
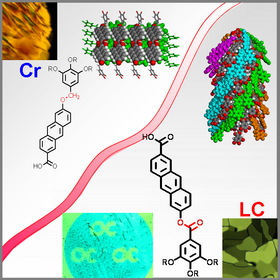
Shape-persistent stars with arms consisting of conjugated oligomers possess empty space which is filled by covalently bound fullerenes and other nanoparticles via spacers to form hybride materials between organic and inorganic materials. These may be a combination of donors and acceptors (dyades). The structures are uncovered by X-ray scattering (SAXS, MAXS, WAXS, GISAXS), modelling (Materials Studio) and X-ray simulation. The optical properties are studied by UV-Vis- and fluorescence spectroscopy. Depending on the core unit also triades can be designed, which are potential interesting materials for photovoltaic applications.
M. Lehmann, M. Hügel, Angew. Chem. Int. Ed. 2015, 54, 4110-4114; A Perfect Match: Fullerene Guests in Star-Shaped Oligophenylenevinylene Mesogens.
German Version: Angew. Chem. 2015, 127, 4183-4187.
Supramolecular Crowded Stars
Space-filling of void in shape-persistent molecular structures forming LC materials is a novel exciting research topic in liquid crystals science. For hexasubstituted benzene derivatives every second substituent is a long arm scaffold generating the free space, whereas the other substituents are small functionalised phenyl building blocks. With this design LC- Endoreceptors are developed, which can uptake at maximum three guests. We study how the guests influence the self-assembly and the physical properties by polarised optical microscopy (contact samples), DSC (phase diagrams), FT-IR microscopy and X-ray scattering. The space is controlled by the length of the conjugated oligomer arms, thus even the size of the guests and their functionality can be adjusted for future applications.
M. Lehmann, P. Maier, Angew. Chem. Int. Ed. 2015, 54, 9710-9714; Shape-Persistent, Sterically Crowded Star Mesogens: From Exceptional Columnar Dimer Stacks to Supermesogens; German Version: Angew. Chem. 2015, 127, 9846-9850.
P. Maier, M. Grüne, M. Lehmann, Chem. Eur. J. 2016, DOI 10.1002/chem.201604505, A Star-shaped Oligo(phenylenevinylene) Liquid Crystal Host with an Anthracene Guest - A Double Nanosegregating Supermesogen
M. Lehmann, P. Maier, M. Grüne, M. Hügel, Chem. Eur. J. 2016, DOI 10.1002/chem.201603611, Crowded Star Mesogens: Guest Controlled Stability of Mesophases from Unconventional Liquid Crystal Molecules.
Semiflexible Stars with Stilbene, Acene and Pyrene Chromophores
Semiflexible oligobenzoate stars fold to various conformers in order to optimise the packing into liquid crystal phases. Thus E- and cone- shaped conformers are formed and self-assemble in lamellar, columnar, cubic bicontinous and cubic micellar phases. The preorganisation, for example in four-arm stars, is used to control the folding and generates Janus-type liquid crystals with stilbene, acene or pyrene moieties showing host-guest chemistry with electron deficient chromophores leading to novel LC structures.
M. Lehmann, ”Star-Shaped Mesogens” in Handbook of Liquid Crystals, 2nd edition, Vol. 5, Chapter 5 (eds. J. W. Goodby, P. J. Collings, T. Kato, C. Tschierske, H. Gleeson, P. Raynes), ISBN 978-3-527-32773-7, Wiley-VCH, Weinheim, 2014.
M. Lehmann, S. Gloza, S. Roth, Chem. Mater. 2015, 27, 8181-8184, At the Limits of Liquid Crystallinity: Stimuli Responsive 3D Columnar Liquid Crystals and Soft Crystals of Supramolecular Anthracene Mesogens.
Solid-State Assisted Synthesis
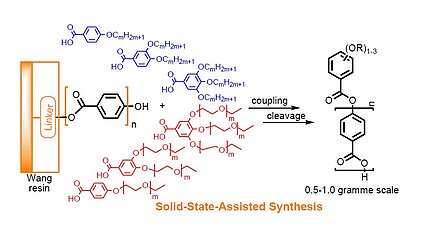
The control over the formation of mesophases on a nanoscale is achieved by nanosegregation of various building blocks and also by the attachment of incompatible flexible chains (alkyl, oligoethyleneoxy, semiperfluorinated chains). The change of the number and especially the nature of chains require a completely new optimisation of all isolation procedures, which is often challenging. The solid-state assisted synthesis introduces the chain-building blocks in the final step and purification has to be done only once after the cleavage from the resin. It is therefore often superior over the conventional liquid phase synthesis, especially when building blocks, such as oligoethyleneoxy chains, have to be introduced.
M. Lehmann, K. Bahndorf, Michael Ohnemus, Steffi Gloza, Sabine Roth, Liq. Cryst. 2015, 42, 1444-1449, DOI: 10.1080/02678292.2015.1056264; Solid-State Assisted Synthesis of Oligobenzoates.