Photoionization with synchrotron radiation
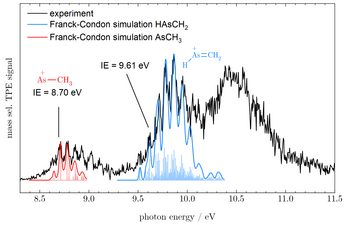
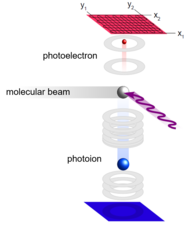
A synchrotron is a broadband radiation source that is used in many physical, biological and chemical fields. In our group, the vacuum-ultraviolet (VUV) range of this radiation is used for structural analysis, investigation of reaction kinetics and analysis of dissociative photoionization of reactive intermediates in the gas phase. To this end, the synchrotron radiation is tuned until the ionization energy of the molecule is reached. This creates a photoion and a so-called threshold photoelectron. This electron has initially no kinetic energy, but is accelerated towards an electron detector by ring electrodes. The cation travels in the opposite direction through a time-of-flight spectrometer and is analyzed on an ion detector. Further tuning of the radiation produces additional threshold electrons when a resonant transition to a vibrationally excited state of the cation occurs. A threshold photoelectron spectrum can be generated by analyzing the electrons at the detector. Each detected electron coincides with the mass of the respective photoion. Accordingly, a mass-selected TPES can be assigned to each signal in the time-of-flight spectrometer. Quantum chemical calculations yield input data for simulations of the spectra that permit to identify isomers of the same mass using this method.
Currently, the focus is mainly on quantum chemical calculations, as the synchrotron used for VUV radiation (Swiss Light Source in Villigen, Switzerland) will not be operational again until the end of 2025.
Recent publications:
1. Emil Karaev, Dorothee Schaffner, Marius Gerlach, Sira Grätz, Patrick Hemberger, Ingo Fischer
Threshold photoelectron spectroscopy of small organo-selenium radicals
Phys. Chem. Chem. Phys., 2026,28, 315-322
2. Emil Karaev, Dorothee Schaffner, Marius Gerlach, Patrick Hemberger, Ingo Fischer
Photoelectron Spectra of Small Gallium and Aluminum Radicals
J. Phys. Chem. A 2025, 129, 27, 6004–6010
3. Emil Karaev, Marius Gerlach, Katharina Theil, Gustavo A. Garcia, Christian Alcaraz, Jean-Christophe Loison, Ingo Fischer
Photoelectron spectrum of the pyridyl radical
Phys. Chem. Chem. Phys., 2024, Advance Article