Just Published in the European Journal of Inorganic Chemistry
04/24/2017Electronic Influences on the Stability and Kinetics of Cp* Rhodium(III) Azide Complexes in the iClick Reaction with Electron-Poor Alkynes
Authors: Luisa Waag-Hiersch, Jan Mößeler, Ulrich Schatzschneider
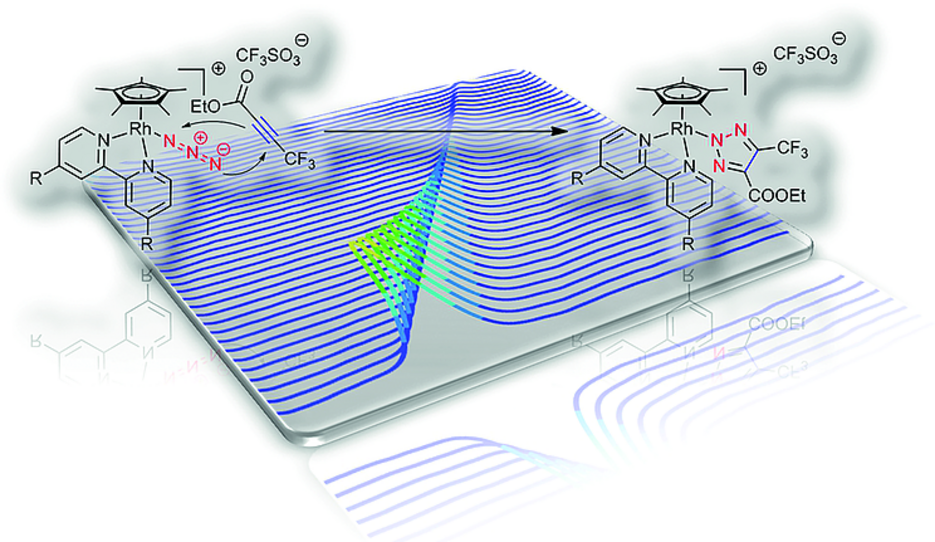
Abstract: Rhodium(III) azide half-sandwich complexes of general formula [Rh(Cp*)(N3)(bpyR,R)]CF3SO3 with R = H, OCH3 were prepared in three steps in an overall yield of 55–65 %. Their stability strongly depends on the 4,4′-substituent on the 2,2′-bipyridine (bpy) ligand and increases in the order COOCH3 < H < OCH3. Consequently, no stable product could be isolated for the complex with the methyl ester substituent. The title compounds easily underwent cycloaddition reactions with ethyl 4,4,4-trifluoro-2-butynoate in acetonitrile at room temperature in the absence of catalyst. The resulting triazolate products were simply isolated by precipitation in 65–75 % yield. The trifluoromethyl group enabled facile monitoring of the reaction by 19F NMR spectroscopy. Kinetic studies with solution IR spectroscopy revealed pseudo-first-order rate constants of 2.4 to 3.8 × 10–3 s–1, and the reactions were accelerated by the presence of electron-donating groups at the 4,4′-position of the bpy coligand. These values are of a similar magnitude as those for the Staudinger ligation utilized for bio(macro)molecule functionalization.
Link: http://onlinelibrary.wiley.com/doi/10.1002/ejic.201700199/abstract